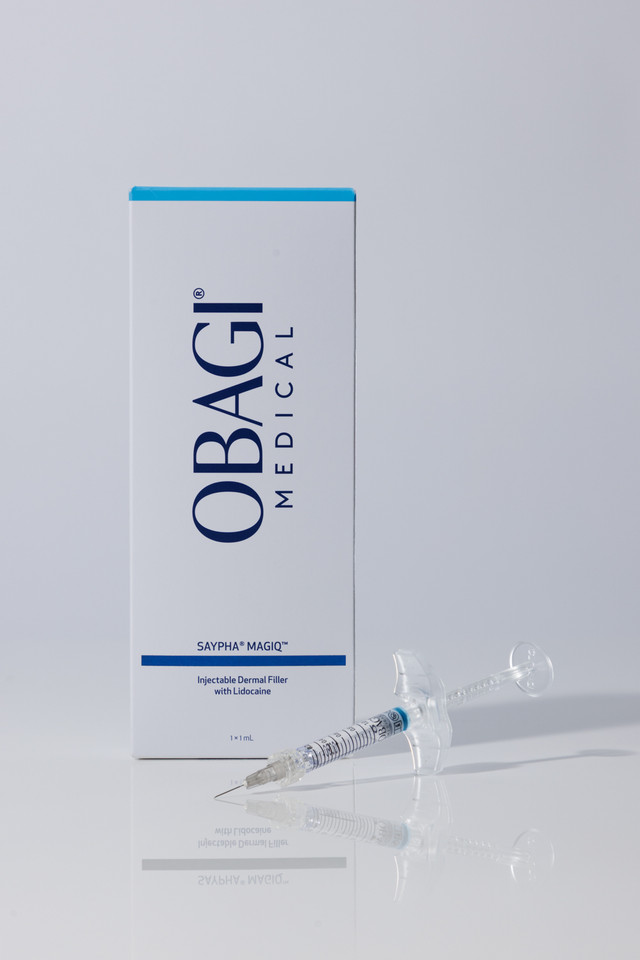
How to Use
Ingredients
Cross-linked hyaluronic acid (in phosphate buffer) with 0.3% lidocaine.
Safety Information
- Prescription use only
- Directions for Use for full administration guidance and safety information are available at obagi.com/saypha
- Approved Use: Indicated for injection into the mid to deep dermis for correction of moderate to severe facial wrinkles and folds (such as nasolabial folds) in adults over the age of 21
- The most commonly observed side effects include swelling, redness, pain,bruising, tenderness, lump formation, and itching at the injection site. To learn more about serious but rare side effects and full Important Safety Information, visit obagi.com/saypha
- To report a side effect with any Saypha® product, please call Obagi® Customer Support at 1-888-798-9809
INTELLECTUAL PROPERTY
- For patent information, visit obagi.com/saypha.
- saypha® is a registered trademark of Croma-Pharma GmbH used under license.
- Obagi® and all derivatives are registered trademarks of Obagi Cosmeceuticals LLC.
- All other products/brand names, whether designated by notice or not, are trademarks of their respective owners.
Clinical Data
- In a U.S. pivotal study of 270 participants, MaglQ™ demonstrated non- inferiority to Juvederm® Ultra® XC through 12 months, confirming comparable safety and effectiveness.*1
- At 3 months, 90% of patients were pleased with their MaglQ™ results.*2
* This summary reflects the clinical trial as conducted and published by Croma-Pharma®. Obagi® Medical was not involved in this research, and all information provided originates from Croma-Pharma®.
1. Data on file. FILIDO Clinical Study Report. Croma-Pharma GmbH; 2025.
2. Downie J, Gold M, Joseph J, Green J, Fabi S, Bank D, Cohen JL, Shamban A. Weiss R, Krames-Juerss A.
Monheit G. Multicenter, Randomized Split-Face Trial of a Crosslinked Hyaluronic Acid Filler With Lidocaine for
Nasolabial Fold Correction. Aesthet Surg J. 2025 Aug l:sjafl37